There are several metal surface finishing types for prototyping and production. However, not all of them fit well into the workings of all products. Many surface finishes focus on improving the aesthetics of your product. Others tend to improve the product’s functionality better, while some do both. In this article, we will focus on metal plating finishes.
Many people confuse metal plating for metal coating, and this guide will help you tell the difference. A plated finish offers several benefits to the material, and there are various reasons for choosing to plate. Here, you will learn the various types of plating techniques and their effects on the metal surface. The tips you get here will also help you ensure improved finish quality.
What Are Metal Plating Finishes?
Let’s go back to the basics! What is metal plating? The plating process is a post-production process. It involves the coating or covering of the surface of a workpiece with a thin layer of metal.

The basic understanding of plating is to have a thin layer of one metal coating a substrate. Consequently, the aim is to improve the overall quality of the product. Some of the many benefits of metal plating finishes include:
- Improving corrosion resistance
- Hardening the material surface
- Improving paint adhesion
- Boosting solderability
- Improving wearability of part
- Reducing friction
- Altering conductivity and conductibility
- Improving heat resistance
- Providing shield against radiation
The uses of plating differ in precision machining because it can change the qualities and performance of materials.
Metal Plating Finishes: The Basic Principle
Still on the basics, let’s discuss the basic principle of the plating process. Plating comes with many steps and needs some considerable steps from pretreatment to post-treatment.
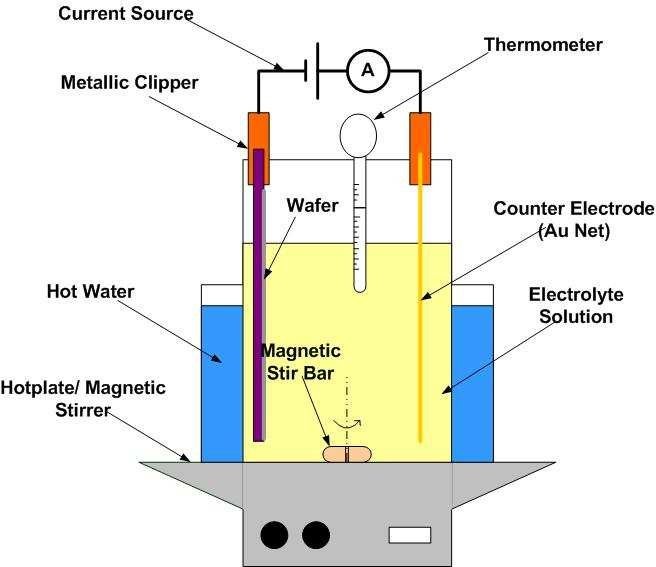
You need to know that there is a wide range of plating techniques, which we will cover in this guide. However, for this section, we shall focus on the basic procedure for the most popular plating process. That is electroplating.
Step #1: Metal Plating Finish Pretreatment
The first step to plating is to prepare the substrate. So, you need first to pretreat it to prepare the substrate for metal plating. This is an important process aimed at removing all harmful contaminants, greases, and oils.
These materials may hinder the plating process and will prevent satisfactory and uniform disposition. Depending on your preferred plating or base materials, this step may include several sub-steps. The sub-steps may range from cleaning to rising and other pretreatments.
Once proper finishing of the base material is complete (in cases where there are welds, burrs, and other surface inconsistencies), then you can now clean the substrate. There are several techniques involved in cleaning the substrate’s surface. It can be by running it through basic or acidic chemicals.
The chemical cleaner you use will depend on the kind of material you work with. For example,
- Alkaline cleaners: These are products containing chemicals like carbonates, phosphates, borax, etc. They work better while doing a plated finish cleaning on steel, copper, nickel, stainless steel, lead, and titanium.
- Acidic cleaners: They include hydrochloric acid, sulfuric acid, and several others. They are recommended for cleaning metals like magnesium.
- Inhibited alkaline cleaners: These cleaners help to overcome the problem of negative caustic reactions on zinc, aluminum, brass, bronze, and tin.
Also, you should know that some base materials require only one cleaning. Others may need two or multiple runs of cleaning. After each treatment, thoroughly rinsing the substrate in distilled water will help remove all chemical cleaners.
Step #2: Determine Effectiveness of the Cleaning Process
The requirements of your parts will determine the plating process. At the same time, it determines how clean your substrates should be. For example, some cad plating techniques require only removing dirt and bulk soil.
On the other hand, others require completely removing oil and grease. How then do you check for your substrate’s cleanliness?
- Water break test. Once you completely rise the final product, hold the base material and notice how water pours off its body. If the pouring off is in one large sheet, then there is no oily residue. However, oil and grease may still remain if there’s beading of water.
- Wipe test. Here, wipe the substrate’s surface using a clean cloth. If you notice dirt or residue on the cloth, you may need to clean the substrate further.
- Reflectivity. The reflectivity of some products helps to gauge their cleanliness easily.
Step #3: Set Up the Plating Station
Once you achieve the adequate cleanliness level, you can now begin the process. It states with the assembly of the station. First, you need a rectifier or other source of direct current. Remember, our focus is on electroplating. Other materials include a tank (or barrel), a cathode, an anode, and a proper plating solution.

The anode includes solid pieces of metal you want to deposit, while the cathode is your substrate. Most manufacturers use water as the plating solution. It is fairly easy to set up a plating station. First, Attach the negative lead of your rectifier to your substrate. Then, put the positive lead right in the plating solution.
Step #4: The Plating Process
As soon as the electrical current turns on, the deposition process begins. If you want a thicker plated finish on your product, then you need to expose them longer to the current. You need to consider some variables when for different metals before plating. These variables include voltage levels, temperatures, immersion times, etc.
In some cases, there may be already prepared electroplating solutions. In that case, you will have the settings for each factor printed on the container. Generally, higher voltages tend to deliver more adequate results. That way, there won’t be bubbles in the solution.
Step #5: The Post-Treatment Process
Once you achieve proper deposition on your metal surface, it is often necessary to conduct post-treatment cleaning. This is important to inhibit tarnishing. Several cleaners are available in the market. The electrolytic polishing after electroplating is also an effective technique. This works to improve corrosion resistance.
Step #6: Waste Disposal
There is often the creation of heavy metals after many plating processes. These metals can be hazardous due to their high toxicity levels. So, it becomes important to quickly and effectively. Pretreating the plating wastewater prior to its disposal is an effective way of doing this.
Standard Industrial Plating Finishes
The following are the standard plating finishes available at the industrial level
Zinc Plating

Zinc is one of the most inexpensive materials that provide galvanized coatings on metal substrates. Application of zinc is by way of molten bath dipping and spraying. The substate is the cathode, while metallic zinc is the anode in a soluble zinc salt electrolytic bath. Zinc plating process produces a very ductile coating. For this metal plating finish, it is easy to control the thickness and uniformity.
Chrome Plating

This plating process usually involves using chromic acid and trivalent chromium baths to produce an overlay on the metal parts. The main aim of chrome plating is to improve the aesthetics of the material. However, chromium metal plating finishes also increase the corrosion resistance and hardness of the material. Those chrome-plated parts are suitable for industrial applications. Sometimes, it also helps to restore tolerances on worn parts.
Copper Plating

When your applications call for cost-efficiency and high conductivity, copper plating is the way to go. This procedure usually serves as the leading coating pretreatment for the following plated finish. It is one of the most popular metal plating finishes for electronic components like circuit boards. It is a popular choice because of its low material cost and high plating efficiency.
Nickel Plating

Another popular plating metal is Nickel because of its usefulness in electroless plating. Nickel plating helps to coat household products like cutleries, shower fixtures, doorknobs, etc., to enhance aesthetics and wear resistance. This plating technique is best for aluminum and copper. However, it works on several other metals. It also serves and the underlying plating for chromium.
Gold Plating

Gold is popular for its high electrical conductivity and resistance to oxidation. A simple way to impart these important properties on silver and copper metals is by gold plating. Its application is extensive in improving conductivity in electronic parts like electrical connectors.
Silver Plating

Similar to gold plating, silver plating also improves the aesthetic appeal of the material involved. Electrical conductivity also comes to play when it comes to silver plating. Many manufacturers choose silver plating for its cost-effectiveness as it is cheaper than gold. It also plates parts made from copper more adequately.
Different Metal Plating Techniques
There are many ways to impart metal plating finishes on metal parts. Here, we will examine the various types and their effects on the material.
Electroplating Metal Plating Finishes
As mentioned earlier, electroplating is one of the commonest methods of metal plating. This method uses an electrical current in a chemical solution to dissolve ions (charged metal particles).

In the process, the charged (positive) metal ions get attracted to the part in question. The part, in this case, is the side with the negative charge on the circuit. When you place the part in the chemical solution, the dissolved metal particles rise to the material’s surface.
The end result is a smooth, rapid, and even coating on the plated material. This effectively offers protection and a decorative look to the metal part. Electroplating also improves the physical, chemical, and mechanical properties of the part, affecting its behavior during machining.
Electroless (Autocatalytic) Metal Plating Finishes
As the name implies, electroless plating does not use any external electric current. Instead, it involves the induction of metal atom reduction through a chemical reaction. That is, the solution of metal particles converts to a metal solid when you mix it with a reducing agent.
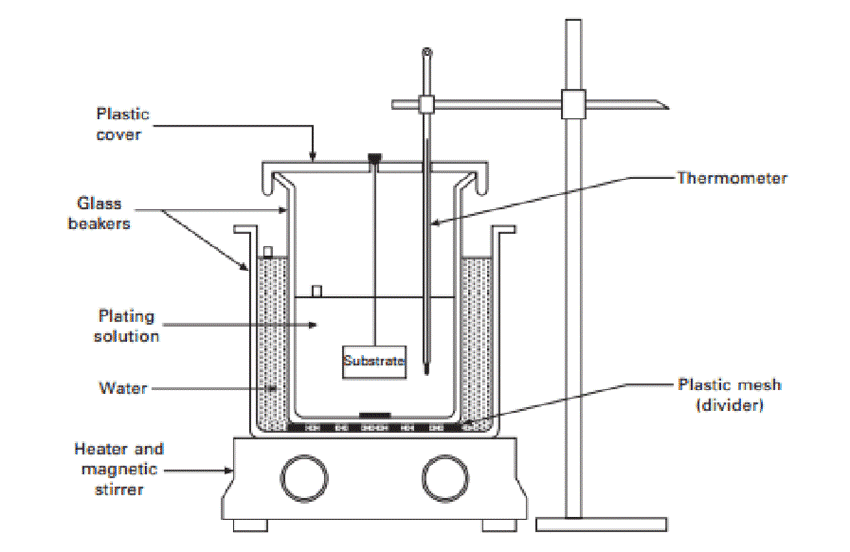
The result of electroless plating is the solid plating metal overlaying the material. Electroless plating confers a fine plated finish on a wide variety of materials. This type of plating is also more cost-effective because there is no external electricity or plating baths required.
However, it is a slower process. It also can’t make thicker plates, and you may find it harder to control than electroplating. However, it proves to be an effective plating method for nickel plating. This metal touch plating offers protection advantages, alters the solderability and conductivity of the material.
Immersion Metal Plating Finishes
Immersion plating involves the dipping of metal into a solution containing metal ions of a noble metal. The ions of the noble metal are usually more stable. Therefore, a natural ‘pull’ occurs to displace the metal ions from the initial metal to give a thin layer of the noble metal ions.
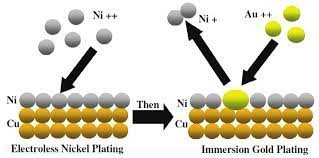
Immersion plating is quite slower than the initially discussed plating processes. It also only comes into the discussion when we mention noble metals like gold, silver, or platinum. The result of immersion plating is thin plating coverage.
Pros and Cons of Metal Plating Finishes
Plating is an effective post-processing process that offers several benefits. However, it comes with its downsides. Here, we will examine the advantages and otherwise of metal plating.
Pros
- Can plate a wide range of materials
- It has a wide range of specific and architectural finishes
- It is a cost-effective finishing technique
- Offers protection against wear and corrosion
- Provides increased hardness and strength to the material
- Increase the decorative appeal of the material
Cons
- Subject to chipping and cracking in some wearing environments
- It can be quite lengthy
- It may be marked by machining defects if not handles carefully
Applications of Metal Plating Finishes
Metal plating finishes offer several different advantages in multiple industries. Their ability to offer parts with improved strength and hardness makes them useful in the automotive and aerospace industries.

Improving conductivity and corrosion resistance makes them useful in the medical industry and making tools and optics. Some of the common applications include:
- Chrome plating for car parts, bath taps, wheel rims, etc.
- Zinc plating is useful for iron parts in automobiles and bridges.
- Electroplating tin onto iron makes good food storage parts like tin cans.
- Silver or gold plating is valuable in the jewelry industry.
RapidDirect: Your Metal Finishing Solutions Experts
As you begin to evaluate metal plating finishes for your applications, you should search for experience and expertise. The best company that offers a perfect mix of both is RapidDirect. RapidDirect has long years of experience in offering cost-effective surface finish solutions for a wide variety of industries.
We offer an extensive array of materials and finishes, including metal plating. Our team of experts works with you to create a custom metal finishing procedure. This way, you can be sure of high-quality services, reduction in operating costs, and overall satisfaction. Upload your design file today, and let’s get to work!
Considerations for Metal Plating Finishes
Materials with a plated finish offer many benefits, ranging from aesthetics to improved functionality.

However, you need to understand some factors to help you reap the benefits of this process. Here are some of the factors you must consider before beginning the plating process.
Pre-plate material conditions
Before plating any material, such material must be in a plating-ready state. For example, a substrate with oil buildups, oxides, heat treatment scales, and other residues may have the plated deposit failing to adhere to its surface. Therefore, you must remove these buildups using pretreatment methods. These methods include chemical descalers, acid pickles, deoxidizers, alkaline presoaks, vapor blasting, etc.
Thickness tolerance
Excessive or insufficient coating thickness is likely to cause plating problems. The desired reality is to determine the adequate range of desired maximum and minimum thickness. Keep the following in mind to make the range specific and realistic in your metal finishing request.
- Electroplating results in even coatings. In this case, tight thickness tolerance will not be an issue.
- Electrolytic plating causes settling of finishes on some areas more than others. For instance, plating deposits tend to become thinner within ¾ inches of a recessed area or a corner. So, tight range tolerance is not advisable for flat and simple shapes.
- Specify the surface areas on a product that needs to remain within a tight plating thickness. It is important to consider corners, recesses, and other geometric factors.
- Establishing checkpoints to determine plating tolerance requirements is also important. Be sure to have such checkpoints on surfaces where it makes sense.
Surface roughness
You must also consider the texture of the product’s surface prior to plating. The surface roughness chart is an outstanding guide to help with this. Handing over products with smoother surfaces will more likely give a better result after plating. However, you should note that the exact results depend on the metal used in plating due to different leveling properties.
Hardness or strength specifications
Most metal plating finishes offer increased hardness and strength to the surface of products. Therefore, it is essential to note the current strength and hardness properties of the component. Then, your chosen plated finish should work towards meeting the specifications as set out in the initial design.
Thread inclusion
It will be best to consider other aspects of thickness for products that include any machining thread. Plating parts like screws, hydraulic fittings, etc., are often about four times thicker than flat surfaces. So, you must factor the buildup into your choice of plating process. This will ensure that the threaded parts can meet important specifications and fit together.
Metal Plating Finishes FAQs
Electroplating is the commonest plating process. This method involves dissolving positively charged metal particles using an electrical current in a chemical solution. It works by a theory of hydrolysis to deposit one metal over another.
The key difference between these two metal finishing methods is that you can carry out coating on both non-conductive and conductive surfaces. However, you can only carry out a plating process on conductive surfaces.
Electroplating is a versatile finishing technique that offers advantages in several industries—for example, zinc electroplating help to confer a plated finish on iron parts in automobiles. Also, chrome plating is suitable for car parts, bath taps, etc.
Conclusion
Plating processes should be simple and effective. After adding plated finishes to your material, you can be sure of improved aesthetics and functionality. Plating also improves the chemical and physical properties of products. Therefore, it is vital to work with an experienced plating company. Contact RapidDirect today, and let’s make your experience a seamless one.


